 As a board-certified rheumatologist at Boston Medical Center and researcher specializing in scleroderma, Andreea Monica Bujor, MD, PhD, is always looking to improve the lives of those with this rare autoimmune rheumatic disease. For years, she wondered if there was a better way to diagnose early skin changes in scleroderma patients. That quest led Bujor and her team at the Boston University Chobanian & Avedisian School of Medicine to explore an innovative technique called spatial frequency domain imaging (SFDI).
As a board-certified rheumatologist at Boston Medical Center and researcher specializing in scleroderma, Andreea Monica Bujor, MD, PhD, is always looking to improve the lives of those with this rare autoimmune rheumatic disease. For years, she wondered if there was a better way to diagnose early skin changes in scleroderma patients. That quest led Bujor and her team at the Boston University Chobanian & Avedisian School of Medicine to explore an innovative technique called spatial frequency domain imaging (SFDI).
Scleroderma causes the body to produce excess collagen, leading to fibrosis in the skin or internal organs. There is no cure, and for some with severe disease, the condition can be life-threatening.
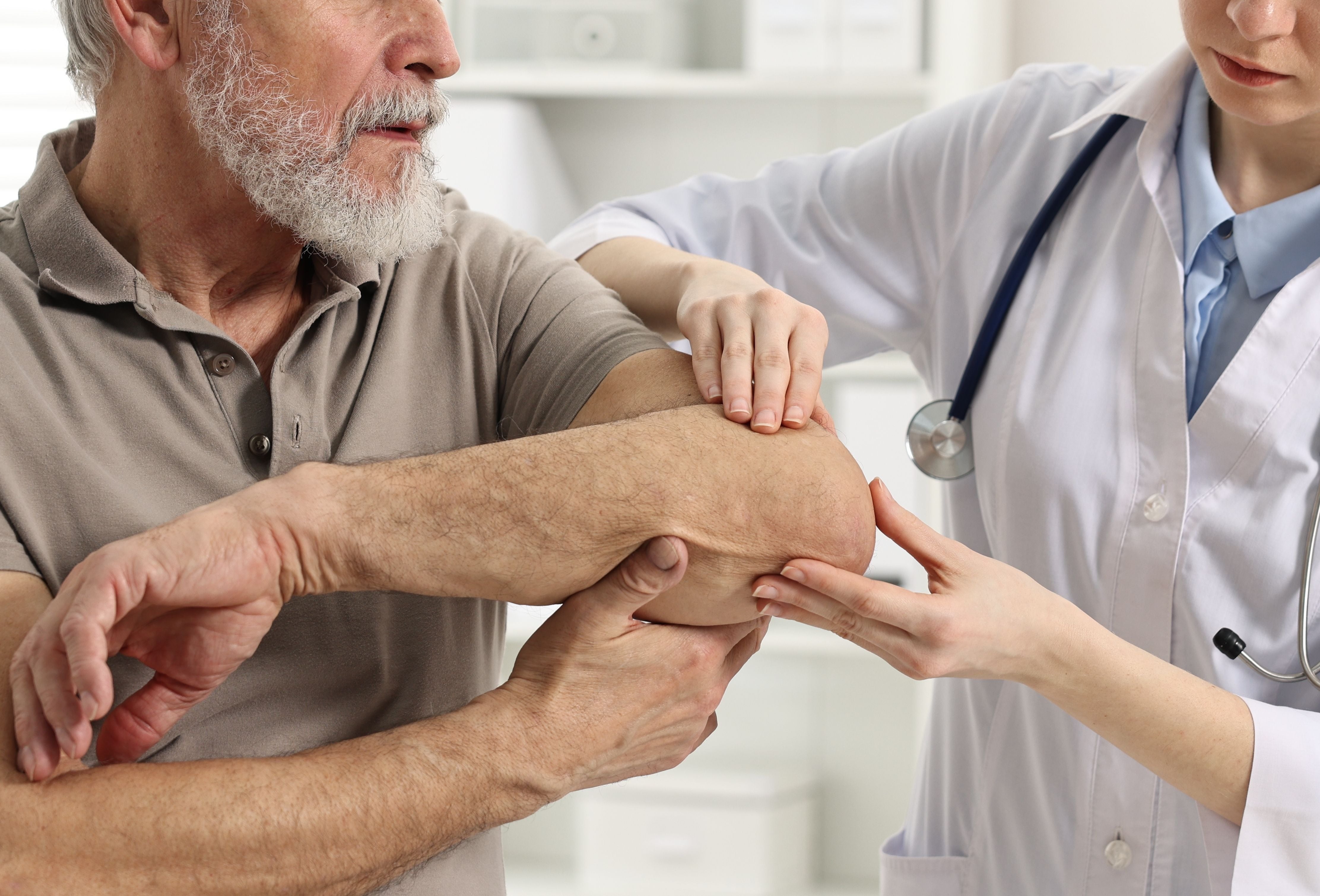
The current standard for assessing skin thickening is the modified Rodnan skin score (mRSS), which involves pinching the skin with two fingers to feel for thickness in 17 areas of the body. A score of 0 for an area means the skin is normal, while a score of 3 indicates severe thickening. The total score helps physicians determine disease severity and progression.
“As you can imagine, this technique is not very objective. It depends on the physician that is performing the test, including their experience and their training,” says Bujor. “Our research project is validating SFDI, a new technique that can detect skin changes in scleroderma patients earlier and more objectively. This is really exciting because it will help patients get diagnosed earlier and receive treatment earlier.”
An Innovative Collaboration with Biomedical Engineering
To create the technology, Bujor and her team collaborated with Darren Roblyer, PhD, an SFDI expert in the Biomedical Engineering Department at Boston University. “We started talking before COVID to see if a device he had created could pick up changes in scleroderma,” explains Bujor.
In this technique, they use near-infrared light at different spatial frequencies and wavelengths to illuminate the skin of patients. A camera captures the optical properties of the skin from a surface of up to 10 cm by 10 cm. Whatever gets absorbed and reflected back is captured by the camera and can be quantified. This technique can sensitively detect skin changes related to fibrosis by picking up alterations in the collagen-rich dermal matrix and other optical properties of the skin.
“There are many combinations of spatial frequency and wavelength,” explains Bujor. “So, Dr. Roblyer has been working with our patients and reviewing the data we've collected over the past five years to find the best combination.”
Her team found that the technique can differentiate people with scleroderma who have no obvious skin thickening by the pinching method from people without scleroderma. “We may feel the skin is the same, but the device can tell us with very good precision that there are clear differences between them,” says Bujor.
Creating a Path for Better Treatment Options
So far, Bujor’s team has one published paper on SFDI and is working on a new paper that is set to publish soon. That research was chosen as an abstract and oral presentation at the 2025 American College of Rheumatology Annual Meeting. “I’m hoping our research might bring us FDA-approved treatments for the skin, which we don't have right now, despite all the clinical trials,” says Bujor. “Many people feel that the reason clinical trials for skin treatments have failed may be at least in part due to limitations in the current outcome measure, the pinching method.”
Bujor and her team are currently working with Roblyer to create a handheld device that acts like a scanner to check patients’ skin, which will provide results immediately at the point of care.
“We hope to have the handheld device ready within two years, and then plan to distribute it to other scleroderma centers,” she says. “There is a lot of interest in collaborating and in using it as an exploratory outcome measure in clinical trials. Because scleroderma patients are so rare, clinical trials for the disease are always multinational and multi-center, and it's not viable to validate it just in one center,” Bujor explains.
“It was a lot of work, but we did it all here at BMC to be able to validate it to this point. I’m so proud of our team that we were able to do it locally.”
